Case Study: The Clinical Translation of Transcranial Magnetic Stimulation
Key Lessons:
- Dedicating IP to the public domain doesn’t necessarily yield more public benefit than patenting
- Clinical trials for depression are hard to run
Treating depression with magnetic fields sounds, to most people first hearing about it, about as legitimate as treating depression with healing crystals.
But in fact this technique, called Transcranial Magnetic Stimulation (TMS), is well-established, notoriously safe, and fully FDA-approved.
The saga of how this came to be is worth studying for anyone interested in neurotechnology.
The Basics
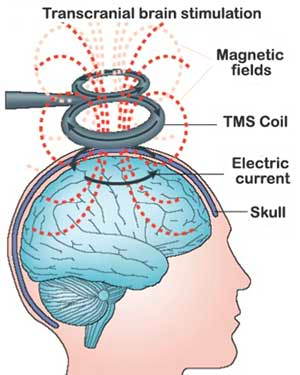
TMS uses electromagnetic induction to create an electrical current in brain tissue. It does this from outside the skull. No surgery required - in fact no physical objects need touch the body at all.
 (Source)
(Source)
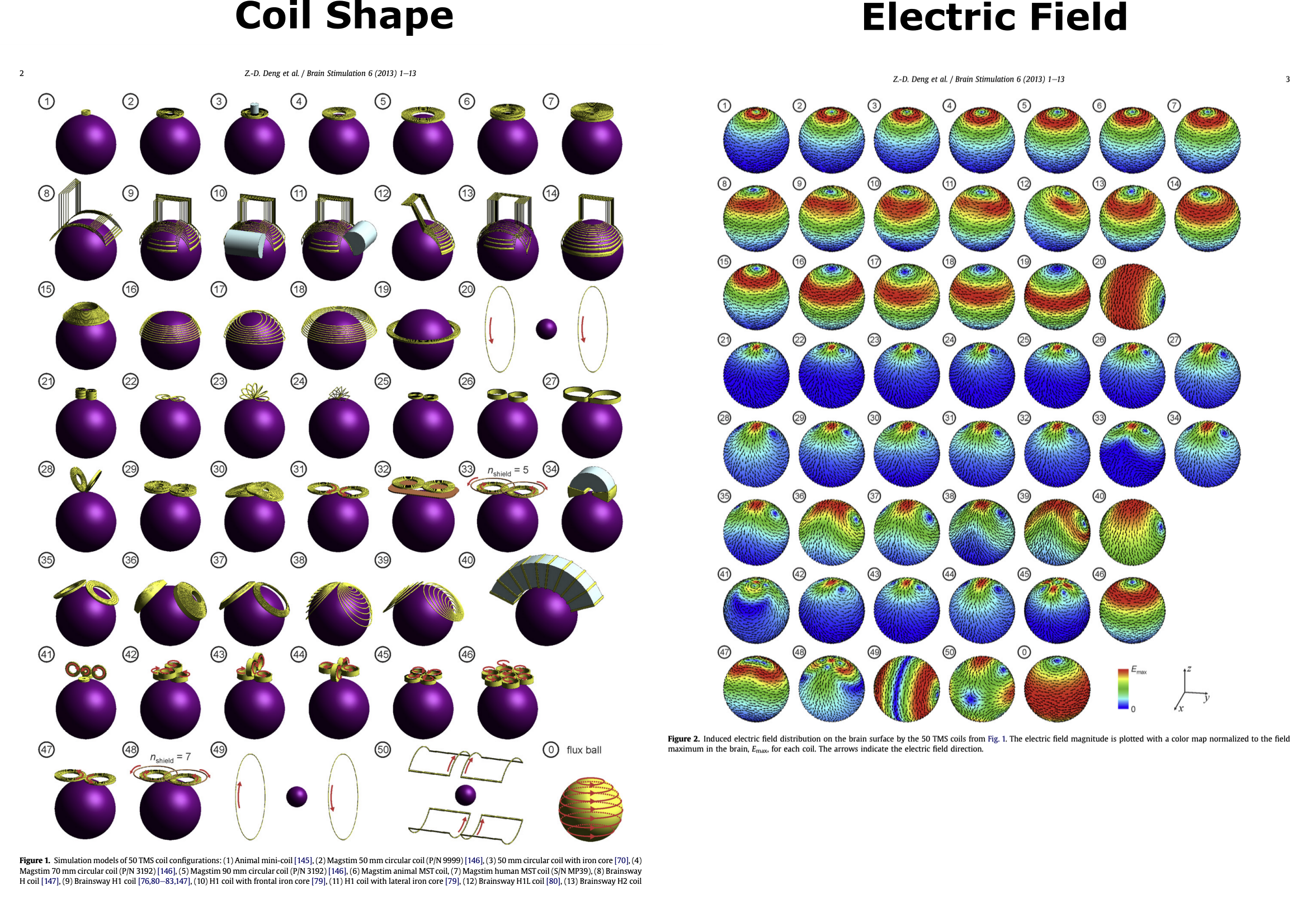
Different TMS coil shapes produce different electric fields in the brain:
 (Source)
(Source)
These days most clinics and researchers use a figure-eight coil, which has a focus in the center of the 8.
TMS induces electrical currents in the brain in short pulses. Single pulses can be used to fire neurons on-demand for neuroscience experiments or diagnostic purposes. But when used therapeutically, patients are typically stimulated with dozens of TMS pulses per second for 30+ minutes a day, 5+ days a week, for 5+ weeks. It’s notoriously noisy.
This is called repetitive-TMS therapy (rTMS, though often just referred to as TMS), and it’s FDA-approved to treat depression, OCD, smoking addiction, migraines, and depression-associated anxiety.
There are other magnetic neurostimulation techniques that use lower-intensity magnetic fields than TMS. But TMS is the most widely used magnetic stimulation method and the only one approved by the FDA.
Early History
People have been putting their heads in magnetic coils for a long time, though not nearly as long as they’ve been running electrical currents through them.
Jacques-Arsène d’Arsonval is apparently the first person to have tried putting people’s heads in a big magnetic field. In 1896 he reported “phosphenes and vertigo, and in some persons, syncope” from the adventure. This effect was independently discovered in Germany and the UK soon after, because apparently reinvention is easier than learning to read French. (Source)
And then in 1914, research on TMS-avant-la-lettre stopped for 30 years, for reasons I don’t know. The timing would seem to implicate WWI, though the last paper on TMS in 1914 was published by researchers at the far-afield University of Washington in Seattle, and all the early TMS authors lived until the 1930s except Thompson, the British rediscoverer, who died in 1916 unrelated to the war.
Though it made a few reappearances after 1946, TMS might have remained undeveloped were it not (reportedly) reinvented again by Prof. Anthony Barker. Barker’s big result (presented in a tiny a 500-word paper)1 came in 1985, when he figured out how make fingers twitch with painless magnetic stimulation of the head.
This was a big deal. Scientists at the time could make fingers twitch by stimulating the fingers directly, and they could make fingers twitch with painful electric current run across the skull, but painless finger twitches from the head were a breakthrough. Modern TMS was born.
rTMS Takes Off
Even though there were preliminary results of TMS improving mood in conjunction with electrical stimulation in 1987, for the next decade after Barker’s result single-pulse TMS was explored as a research tool and diagnostic tool, not as a therapeutic tool.
But by 1991 Pascual-Leone et al. had shown that rTMS could interfere with visual perception and speech. And in 1994 they also found that the excitatory effects of rTMS in cortex could last for minutes after the stimulation stopped. This reminded researchers of the (presumed) effects of electroconvulsive therapy, an effective treatment for depression among other things. Concurrently, researchers had anecdotally noticed that subjects in vision or speech experiments who received TMS stimulation in their frontal cortex sometimes felt their mood change.
From 1994 to 1996 at least 6 clinical studies (not all controlled) reported TMS altering mood and depression. A lot of these studies’ results were probably flukes or the placebo effect given what we now know to be effective stimulation parameters for TMS, but that’s rarely stopped a field (and maybe shouldn’t).
In 1996 a big meeting of scientists was convened to lay out standards for rTMS use. Such standard-setting might in hindsight seem overcautious given that the only adverse effects of TMS that had been observed were scalp pain and very rarely seizures. But the nascent TMS field was probably keen to avoid the cultural immolation that electroconvulsive therapy had undergone in the 1960s. Prof. Mark George, a pioneer in the field, didn’t even patent the treatment due to fear it would damage its reputation. (Barker before him hadn’t patented his coil design either.) George now thinks this lack of IP protection decreased overall public benefit from TMS by reducing industry investment in the technology.
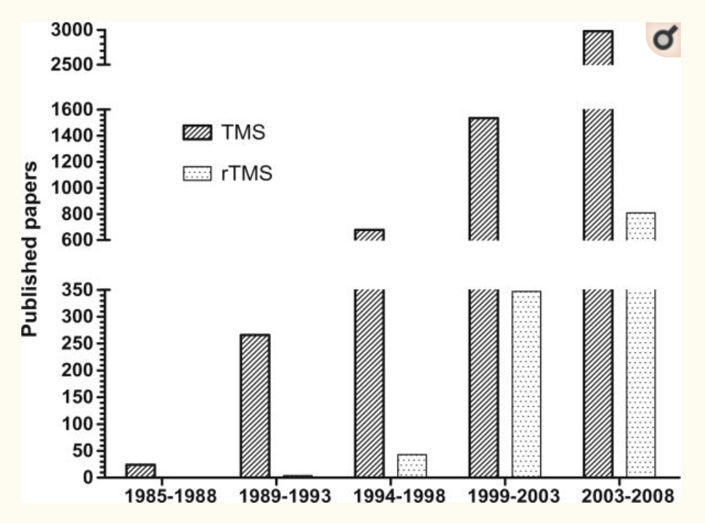
Despite tepid industry interest and the NIH not funding research into it, academic interest in rTMS grew quickly. Here you can see the numbers of published papers mentioning rTMS after the 1996 conference:
 (Source)
(Source)
Most of these papers aren’t clinical trials, but the interest is the point. In fact this plot underestimates how much work was being done on rTMS, since within a few years of the 1996 meeting, clinics and doctors were offering off-label TMS therapy. Supposedly some countries including Australia, Israel, and Canada approved the treatment around the same time as well, though I haven’t found official evidence of this. Also around this time, commercial vendors started selling rTMS devices to researchers, whereas before researchers had been modifying single-pulse TMS systems or building their own.
All of which is a bit depressing.
Why? Because even with the explosion of research after 1996, it would still take another 12 years for the field to develop rTMS into a legitimate therapy for anything. In fact, as discussed below, the field was lucky the FDA approved rTMS in 2008 at all. Not to mention that the approval was very narrow: only for depression in adults who hadn’t responded to one, but no more than one, antidepressant medication. The field would have to wait another decade for TMS to be approved for OCD and smoking cessation.
I can accept the decades of work up to 1996 as the price of serendipity. But as someone who cares about the therapeutic potential of future neurotechnologies, the decades required to develop reliable TMS therapies demand analysis.
What took so long?
One contributing factor to TMS’s slow path to approval was probably the field’s overwhelming focus on depression.
Depression is of course an important and worthy focus of therapeutic development. But developing depression treatments is particularly slow and hard compared to something like migraine, for which TMS would later be approved. Clinical measures of depression - surveys like the MADRS and HAMD or qualitative patient self-reports - are fickle and noisy. Placebo response rates in depression trials are large, from 30% to 50% — that’s a lot for a treatment to overcome. And it takes a long time to see whether a depression treatment is having a durable effect.
The focus on depression seems to have come from the hope that TMS could supersede the then-as-now-controversial ECT, and maybe because early TMS studies reported mood-altering effects. Some in the TMS field pointed out at the time that disorders associated with more focal neural activity like epilepsy or focal dystonia would be easier to treat than disorders with less-well-understood circuits like depression. But TMS still hasn’t been approved for such focal disorders, so no idea whether that’s true.
Despite my disillusion at a 12+-year clinical translation timeline, one could argue that rTMS for depression was actually developed pretty quickly given how long clinical trials typically take to run. It took about 3-4 years to perform and publish a clinical trial on rTMS (not including time to secure funding), which amounts to only about 4 “cycles” of experimentation between 1996 and 2008.
Clinical trials don’t have to be this slow. rTMS treatments of the kind patients were receiving in this era lasted around 4-6 weeks, with follow-up visits after maybe a few months. The rest of the 3-4 year timelines went into patient recruitment, IRB approval, writing and data analysis, and the publishing process (peer review plus time in press). And in fact publishing, which accounts for probably 6-12 months, doesn’t really count, since researchers share unpublished work with each other all the time. The first two are the big slowdowns, and I don’t think they’ve improved over time. They’ve probably worsened. This is infuriating. Anyway, I’m just venting. Back to the story.
Another reason for the slow translation of TMS, though by no means unique to TMS, is the large parameter space the field had to explore. At minimum the following can be varied:
- Coil shape (see picture at the top)
- Coil position on the head
- Pulse intensity (roughly the amount of current change during a pulse)
- Pulse pattern (continual vs. delivered in bursts, and frequency of pulses)
- Session duration
- Time between sessions
- Number of sessions
- Types of patients recruited
- Combination with other medications or therapy
- What the patient is doing or thinking about during the treatment
Plus things can get more complicated with current waveform and direction.
One thing that’s clear in reviews and meta-analyses from the time is that the easier a parameter was to vary, the more it was explored. For instance: almost every study used commercially available figure-8 coils. This isn’t surprising since changing the coil shape is really hard: you have to re-engineer the high-voltage electronics every time you do since the coil impedance will have changed. At the other extreme, pulse intensity and pattern are the easiest to change and were the most varied between studies. Nevertheless, to state the obvious: there’s no necessary correlation between importance of a parameter to therapeutic efficacy and how easy it is to vary.
Even among the easy-to-vary parameters, though, the field narrowed its exploration early on in two key ways.
One was the division of pulse patterns into “high” (>1 Hz) and “low” (<=1 Hz) frequency. Early observations from 1996 suggested that high-frequency rTMS was excitatory and low-frequency rTMS was inhibitory, and that excitation and inhibition could last for minutes after stimulation. At least this is what was observed in motor cortex, where excitation and inhibition could be measured via the impact on finger twitching. The field stuck to this dichotomy pretty strictly, and also frequently suggested that the excitatory and inhibitory effects were associated with long-term potentiation and depression. This conclusion is almost certainly oversimplified, but it seems to have been good enough.
The other early assumption that the field made was that the left dorsolateral prefrontal cortex (DLPFC) should be excited and the right DLPFC should be inhibited to treat depression. This idea goes all the way back to 1995 and was inspired by lesion and functional imaging work. The left DLPFC remains the main target of TMS for depression. It’s what the FDA approved in 2008.
In hindsight these two assumptions seem to have been spot on. The earliest TMS trials for depression from 1995 and 1996 used nearly exactly the same stimulation setup as would eventually be approved by the FDA in 2008. The main innovation in those 12 years was turning up the pulse intensity by ~40% and doing 4-6 weeks of treatment rather than 1. There were also big innovations in the quality of sham treatments during this time. (“Sham” is the medical device world term for placebo.)
It’s hard to know what to make of this, other than maybe wishing trials with sufficient power had been run sooner. Though Prof. George thinks running trials sooner would have been premature, pointing to what happened with DBS rushing from open-label trials to big trials that flopped.
Two big things the field did underrate at the time were the importance of coil positioning and what the patient thinks about during the treatment.
It’s harder than you might think to position a TMS coil over the right part of the brain, orient it correctly, and keep it in place for a half hour.2 The way they used to find a patient’s DLPFC was by measuring 5 cm anterior to wherever TMS made the patient’s thumb twitch. But this isn’t where the PFC is in 1/3 of patients, not to mention the DLPFC. They then started using the 10–20 system, but we know from newer results that this is also pretty inaccurate. Even with modern computers and neuronavigation systems targeting is tricky, and for large trials one has to train technicians/clinicians to target correctly and carefully across lots of patients.
We also know now that what a patient does during stimulation can have a big effect on a TMS treatment. The approved TMS treatment for OCD involves a “Personalized Symptom Provocation” - patients are made to think about something that triggers their symptoms while their brain is stimulated. Between 1996 and 2008, as far as I can tell, patients could do whatever they wanted during TMS treatments: ruminate, read, sleep, etc.
What’s the opposite of a slam dunk?
In 2007, a company called Neuronetics was the first to apply to the FDA for approval of a TMS system for treatment of disease - specifically for approval of their NeuroStar system to treat treatment-resistant depression.
The double-blind, multi-site clinical trial they ran to support their application was the largest that had been run at that time. It took almost 2 years, starting in January 2004, to recruit all 325 patients. The IRBs from all 23 (!) sites involved in the study had to approve the trial.
But the massive investment was worth it, because when the trial finally concluded it…uh…still failed its primary endpoint and arguably all its secondary endpoints too.
And then the FDA approved NeuroStar anyway.
The FDA’s Advisory Committee did justifiably slam Neuronetics for statistical shenanigans when they presented their results. You can read the juicy details in the transcript of FDA deliberations on NeuroStar, Jan 2007. But I think the FDA’s decision was still for the best from a patient risk-benefit perspective. Though the evidence for benefits in the trial were weak, rTMS had (and still has) such a fantastic safety profile that approving NeuroStar and letting post-market studies sort out effectiveness seems preferable to me. And indeed post-market studies have borne this out: TMS works about as well as most depression treatments, which is not that well, but better than no treatment at all.
One thing about the Neuronetics trial that most confuses me is why they didn’t survey patients about what treatment they thought they got in order to test the blind. I have no idea why a trial wouldn’t do this, and doing so might have let them avoid critiques like this:
“36% of the active group compared to only 4% of the sham group complained of [head] pain… When [you adjust for patients feeling pain], p-values for the treatment effect became less significant. In particular, the p-value for the primary MADRS endpoint went from borderline significant to [really bad].” — FDA
To be fair, the FDA never sounds excited about anything. Even in their deliberations on the Pfizer COVID vaccine EUA in December 2020 — the most slam-dunk clinical trial in recent memory — their average tone never exceeded “fairly into it”, and only 17 out of the 22 committee members voted to approve.
But the bottom line is that the NeuroStar approval was pretty far from a slam dunk. Plenty of folks let the FDA know it after the fact, too. And another multi-center trial using very similar stimulation parameters showing TMS had no antidepressant effect was published right after the NeuroStar trial. (Though it was for patients who were also taking antidepressant drugs, whereas the NeuroStar trial was for patients not taking drugs.)
Nevertheless, by 2010, the American Psychiatric Association (APA) had included TMS in its Practice Guidelines. Insurance seems to have taken a little while to come around, but it was mostly covered by 2015, though even today it can apparently be hard to get insurance to cover it.
Recent Developments
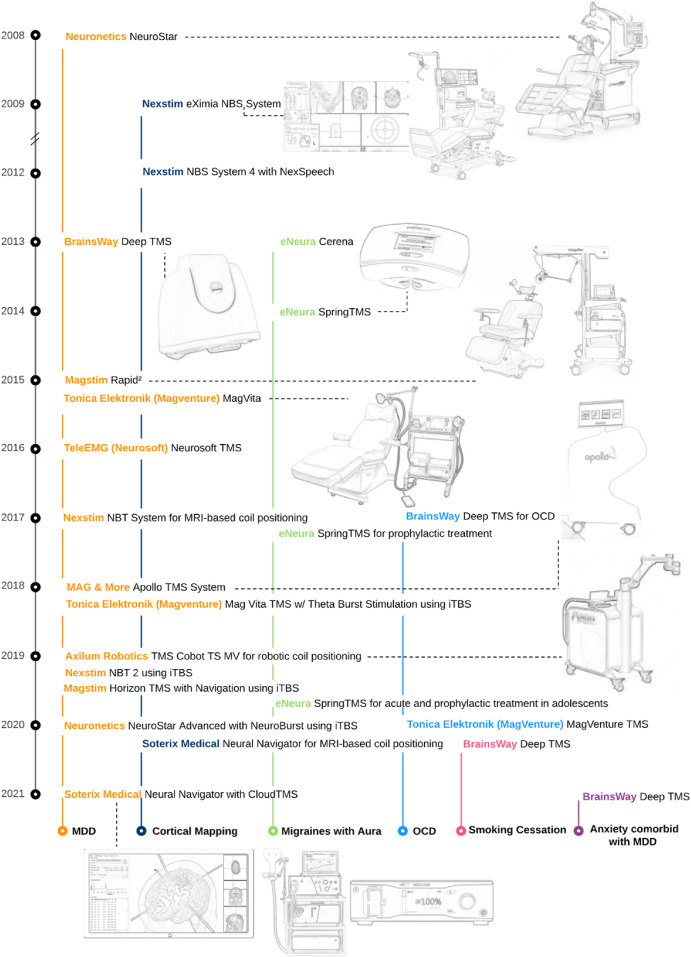
This infographic tells the story after 2008 better than I can:
 (Source)
(Source)
TMS has gone on to be approved by the FDA for migraines, OCD, smoking cessation, and depression-associated anxiety. And TMS is regularly used off-label for a lot of things.
But TMS still has lots of room for growth. The more we can learn from neuroimaging about the cortical circuits involved in neuropsychiatric diseases, the more TMS protocols can be developed to target them. And personalized targeting is showing impressive results that might hugely reduce the treatment time and increase efficacy. If the treatment time can be brought down to just a few days, it’d be hard to argue that TMS shouldn’t become a first-line therapy for depression. It has far fewer known adverse effects than any antidepressant drug — the cost and time are what make it infeasible now.
Let’s just hope we can start measuring this growth in years, not decades.
Have feedback? Find a mistake? Please let me know!
-
Actually 500 words and a picture of the device, which the Lancet told Barker he should include “if it wasn’t too dull.” ↩︎
-
Though at least TMS has undeniable neurological effects (e.g. making fingers twitch) that can be used to calibrate it! Usually the strength of TMS stimulation used on a patient is proportional to how much current is required to make their thumb twitch. Compare that to most tES techniques, where you don’t have any idea where the current is going. ↩︎