Case Study: Transcranial Electrical Stimulation
Key Lessons:
- Similar technologies with different names can elicit wildly different cultural and regulatory reactions
- The number of papers published on a technology, or the age of its field of study, or their number of citations therein, tells you almost nothing about its value or effectiveness
- The US used to have a much stronger DIY-health/treat-yourself-at-home ethic
- The FDA cannot be predicted
- For many indications, if the user is asking whether it’s working, it’s not working
People have been putting electricity through their heads for a very long time. The umbrella term for this is transcranial electrical stimulation (tES).
Every discussion of tES must, by tradition, start with the anecdote that people in antiquity put electric fish on their heads to treat headaches. In the modern era, the most famous types of tES include electroconvulsive therapy (ECT), transcranial direct current stimulation (tDCS), and recently temporal interference (TI). But there are, and have been, dozens of varieties of tES differing in electrode placement,1 waveform, intensity, and duration.
Early Days, ~1900
All due respect to electric fish, tES really got going in the late 19th century with the spread of battery technology. In the “golden age of electrotherapy” in the US around 1900, tES devices nearly identical to modern tDCS devices were “one of the most ubiquitous therapeutic items.” They were sold for anesthesia, nausea, rheumatism, “Female Weakness” (depression?), “Premature Decline in Man” (ED?), and many other ailments, fictional and not. More interesting was that they were sold as much to consumers as to doctors. Self-treatment was considered the norm in medicine until around WWI, and there were strong DIY and direct-to-consumer (D2C) markets for tES. (Source)
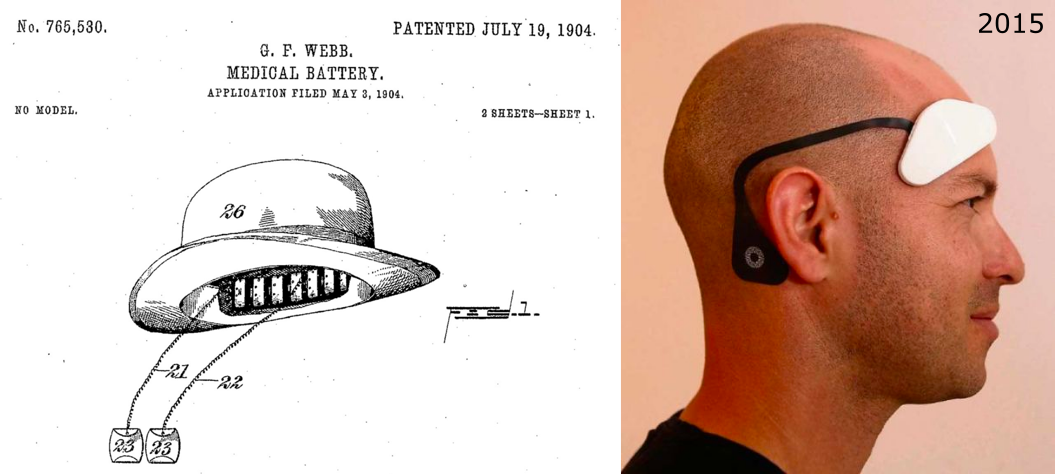
The existence of this “golden age” is a bit surprising given that cocaine and opiates were legal at the time. I’d have thought over-the-counter heroin and similarly potent drugs would obviate consumer demand for tES. Equally surprising is the golden age ending after 1910, since that was when the US government began cracking down on drugs.2 tES remained unregulated and could have filled that void, but didn’t. Explanations include the rise of psychology, DIY healthcare giving way to the professionalization of medicine, and X-Rays stealing the spotlight.
ECT, 1930s+
tES stayed unpopular from 1910 until the 1930s when Electroconvulsive Therapy (ECT) was developed. ECT, also known as electroshock therapy, involves passing large currents through the skull in order to induce seizures. In the spirit of More Dakka, we’re talking 800 mA compared to the <10 mA in nearly all other forms of tES past and present.
In the early days of ECT the treatment was given to conscious, unanesthetized patients, which was obviously quite unpleasant. By the 1950s the field had gotten around to putting patients under general anesthesia during ECT, resulting in a calm, painless procedure. But the cultural damage was already done. Ken Kesey’s depiction of unanesthetized ECT in One Flew Over the Cuckoo’s Nest, and later the film depiction of the same, is often blamed for the huge decline in the use of ECT after the 1960s. (This is what ECT looks like these days.)
The PR failure of ECT is sad, because ECT is among the most effective treatments for depression, though not necessarily long-lasting. The most common side effect is transient memory loss. While it has slowly crept back into popularity over the years, one wonders why it wasn’t able to rebrand itself, especially given how many other tES techniques managed to do so.
Weak t, 1960s+
As ECT receded in the 1960s, lower-current methods began to advance again.
Electrosleep was the first. Electrosleep was the name of a tES method for insomnia where pulses of low currents (<25 mA, usually <10, and often <1) are passed across the scalp. Invented in 1902, it would have died with the other golden-age tES methods after 1910 had it not been kept alive by research in Russia and Europe.
After reemerging in the US in the 1960s, scientists concluded it wasn’t actually doing anything in the brain directly to cause sleep. It was just a relaxing, electric scalp massage. Due to this it was renamed Cranial Electrostimulation Therapy (CET). And then renamed Transcerebral Electrotherapy (TCET). And then renamed Cranial Electrotherapy Stimulation (CES). When I said I was confused why ECT couldn’t rebrand itself, this is why. (source)
There’s a whole saga of Electrosleep/CET/TCET/CES/scalp-massage-with-a-battery trying to get better treatment from the FDA, but it’s not worth going into unless you really want to get in the weeds of FDA device classification.
Also in the 1960s people started playing around (again) with non-pulsed, DC stimulation, but it fizzled out. And in the spirit of scalp-massage-with-a-battery, more work began to be done targeting cranial nerves for pain and headache relief. And by “work” I mean “scientists with the bravery of a bygone era sticking needles into their own skulls to test their ideas”. This line of research continued to the present day with devices like Cefaly, which is FDA cleared for migraine.
But really not much happened until 2000.
A Thousand Flowers Bloom, and the Return of DIY, 2000+
The modern resurgence of tES began with what were almost certainly two false conclusions.
In 1998 and 2000, two groups reported modulating cerebral excitability with the DC tES method what would eventually become known as tDCS. Based on what we know now about how little current gets through the skull and how tiny the currents they used were (<1 mA), there’s almost no chance they were directly changing cortical behavior.
Nevertheless, low-output (<10 mA maximum average current) tES began to rise again. New methods like HD-tDCS, transcranial Alternating Current Stimulation (tACS), transcranial Random Noise Stimulation (tRNS), and transcranial Sinusoidal Direct Current Stimulation (tSDCS) began popping up. The hottest new tES method as of writing is probably Temporal Interference (TI). Originally proposed by Soviet scientists in 1967, it was resurrected/rediscovered in 2017 as a way to potentially get current into the deep brain.
But the most interesting part of the low-output tES resurgence isn’t the methods themselves, but the DIY culture that sprang up around them.
Prof. Anna Wexler tells this story better than I can. But in short, in 2006 a few articles about tDCS make it into the popular press. From there it gets its first Wikipedia article. It’s on Shroomery by 2007. Then on rotten.com. By 2011 it’s got its own DIY subreddit.
D2C tDCS kits start being sold in 2012. By 2014 there were two low-output tES startups: Halo and Thync. Both are still around, and have been joined by many others. While a much smaller percentage of people self-treat with tES now than in 1900, the DIY/D2C tES movement remains strong.
Which raises the question: where is the FDA in all this?
Interlude: How medical devices are regulated in the US
- Since 1976, if you want to sell a medical device in the US, the FDA has to approve it.
- The FDA decides what counts as a medical device based on its “intended use”.
- Teeeechnically, a medical device is any man-made artifact that is intended to impinge upon a human in any way (and isn’t a drug). (21 USC 321(h)(1))
- But since that definition is absurdly broad, the FDA mostly just pays attention
to products that explicitly claim to have a medical purpose, though implicit claims can count too.
- Famously, a dumbbell is both a medical device and not, depending on the ad copy. Same with EEG.
- Wellness aids are a notorious gray area. Safer to market your device as a sex toy.
- There are 3 classes of medical device.
- Class I are low-risk, like tongue depressors. 47% of devices are Class I.
- Class II are moderate-risk, like ventilators. 43% of devices are Class II.
- Class III are high-risk, like pacemakers. 10% of devices are Class III.
- All new devices are automatically Class III until the FDA says otherwise.
- You can petition for your device to be reclassified.
- The higher the class, the more work you have to do to get FDA approval.
- Here’s a flowchart for the whole process.
- Class III (“PMA”) approval is way harder than Class II, requiring years and millions of dollars. People work hard to get reclassified down.
- But, if your device is similar enough to an existing Class I or II (or sometimes III)
device, you don’t have to go through the whole approval process again. You can just prove “substantial equivalence” to the existing device.
- This is called a “510(k)”.
- Protip: if you see a device that’s “FDA Approved”, it went through a PMA. If it’s “FDA Cleared”, it went through a 510(k).
- Devices are also regulated by other agencies, even if the FDA doesn’t consider yours a medical device.
- The FTC takes action against “hazardous…products…without adequate disclosures”.
- The CPSC takes action against “unreasonable risks of injury”, though not in the FDA’s remit.
- The FCC regulates devices that emit RF signals.
The FDA has cleared a number of medium- and high- (>10 mA maximum average current) tES devices, like the Cefaly device mentioned above and ECT devices. But D2C and DIY low-output tES companies have so far avoided language that would lead the FDA to classify them as medical devices. The field has thought about what it would take to get devices FDA cleared or approved. And some of these companies have sought guidance from the FDA and even done clinical trials. But none have yet sought approval for big indications like depression.
Is this because they can’t demonstrate effectiveness? Because goals like cognitive enhancement aren’t considered therapeutic indications by the FDA? Because clinical trials cost too much and there’s likely not much chance of insurance reimbursement to pay them off? Probably a bit of all of these.
To date, neither the FDA nor FTC has made sweeping moves regarding tES devices. Just issued some guidance. Some expect a big reckoning to come for the D2C tES market after the FDA has some bandwidth post-COVID. The way this would probably happen is them saying, “Hey, we the FDA have decided that the way you’re describing these devices makes them medical devices. And we haven’t given you permission to sell them, so shut it down.”
But it’s not in the FDA’s jurisdiction to actually outlaw DIY tES, or any neurotech, even if they wanted to. In fact, it’s not really in any government agency’s. If the government wanted to treat DIY neurotech the way they treat DIY cannabis, they would need to pass legislation like the Controlled Substances Act. If someday soon companies start selling neurotech devices D2C that are, in some sense, as “potent” as cannabis…that’ll be an interesting day.
“What did we learn, Palmer?”
You may have noticed I’ve been avoiding the question of whether any of these methods work for anything.
Besides ECT, which obviously works for depression, schizophrenia, and other indications, the answer is that I don’t know what tES methods work for what purposes, and neither does anyone else, really. Even in 2021, most tES conferences still include a healthy number of papers asking, “Do these methods actually do anything?”
When a neurotechnology is a credence good, like a treatment for stroke rehabilitation, Alzheimer’s, or catatonia, it’s worth resolving such confusion over efficacy with careful study. And of course establishing safety is critical.
But many if not most neurotechnologies are not credence goods. They’re experience goods: goods for which if the user is asking whether it’s working, it’s not working. If you can’t detect an effect in yourself, you either aren’t experiencing one, or you’re not keeping track of your own mentation, mood, behavior, or performance enough for the technology to matter.
The inverse of course isn’t true. If a user says a technology is working, it might be working for reasons we don’t care about like the placebo effect or regression to the mean. And subjects’ desire to please researchers with self-reports should not be underrated. Considering how hard it is to perform tDCS treatments correctly on one’s self (despite their seeming simplicity), it would not be surprising if the entire DIY tDCS movement was fueled by the placebo effect.
Even though this has no real downside given how cheap and safe tDCS devices are, the popularity of DIY tES without undeniable efficacy is something anyone interested in neurotech should reflect on carefully.
Have feedback? Find a mistake? Please let me know!
-
A choice of electrode placement is known as a montage, from the French for “set-up”. ↩︎
-
Even going so far as to try to take the caffeine out of Coca-Cola in the hilariously titled case United States v. Forty Barrels & Twenty Kegs of Coca-Cola. You read that right: the defendant in this lawsuit was barrels of soda, not the Coca-Cola Company. ↩︎